Subscribe
Subscribe today to receive CGD’s latest newsletters and topic updates.
All Commentary
Filters:
Topics
Facet Toggle
Content Type
Facet Toggle
Blog Type
Facet Toggle
Time Frame
Facet Toggle
Blog Post
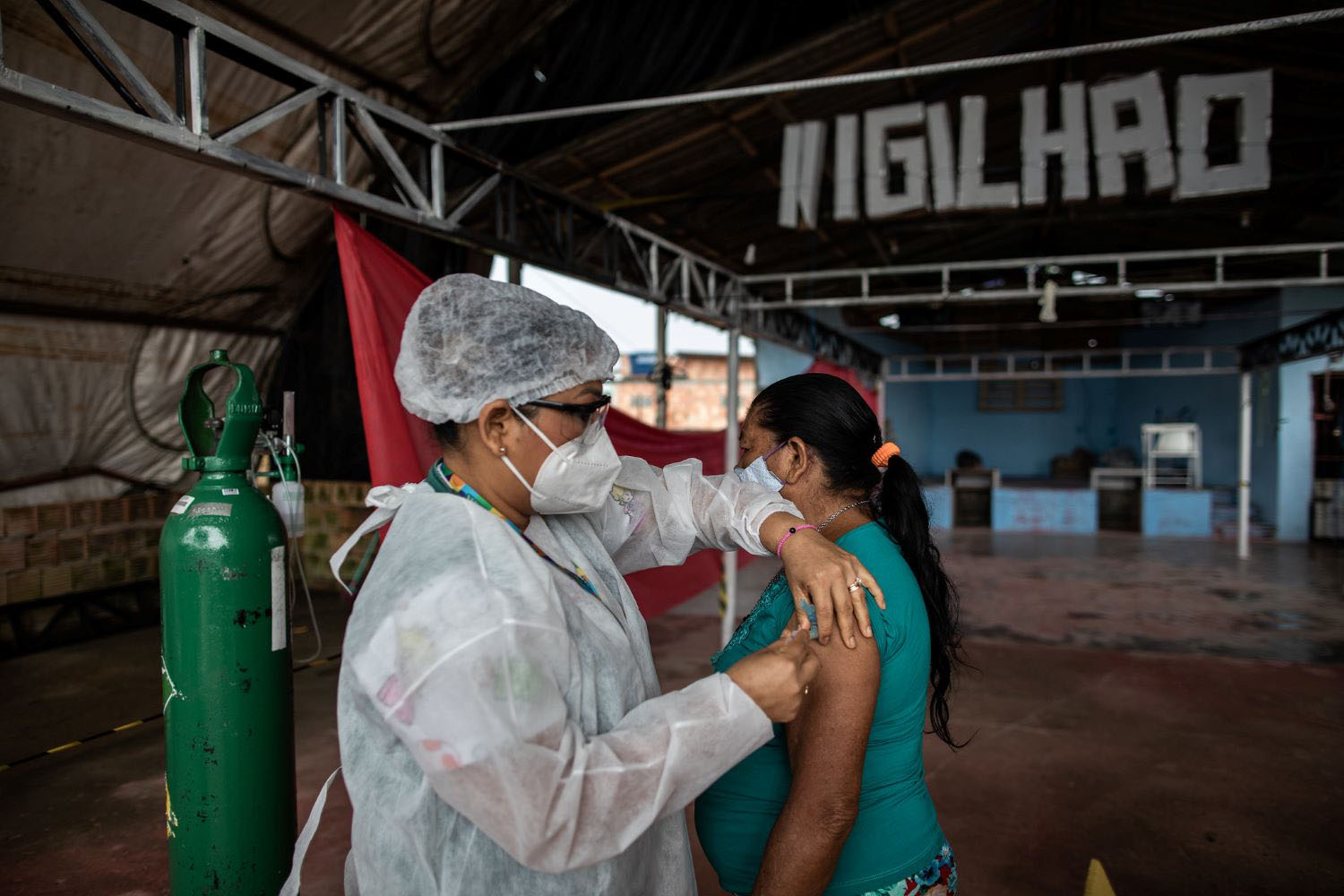
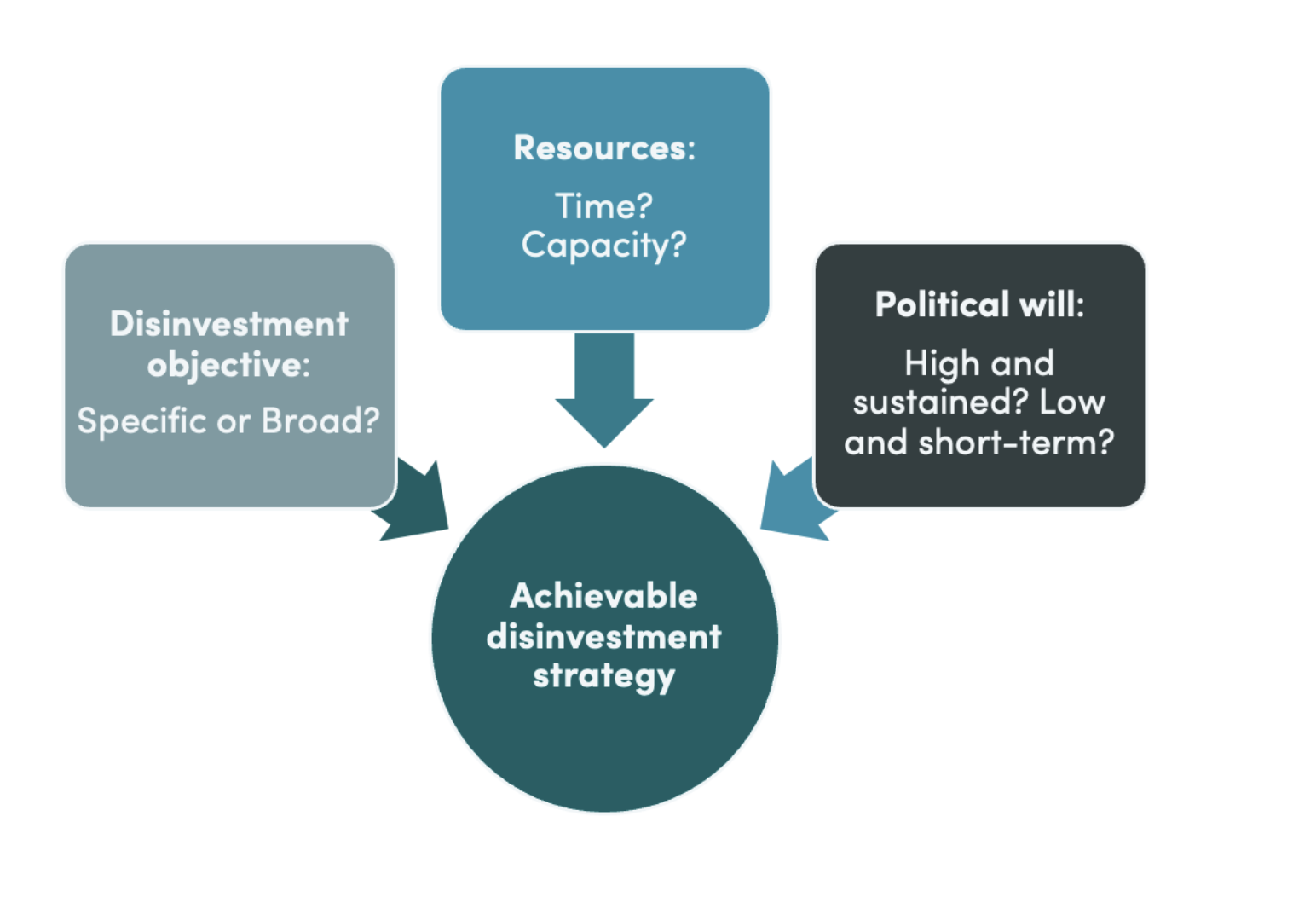
April 15, 2024
In theory, disinvestment offers the potential to release resources for achieving UHC during challenging fiscal times. However, in practice, it is time- and resource-consuming and results in less benefit than expected at the outset. The opportunity cost of launching an initiative needs to be weighed ...
Blog Post
April 11, 2024
There are currently two prevailing methodologies for calculating PCM—the MDB approach and the OECD approach. Both approaches have developed over time and contributed significantly to better understanding of the ways in which private capital is mobilised for development. However, they have key defici...
Blog Post
April 11, 2024
How schools are managed––things like budgets, staffing, and planning––matters for school effectiveness and children’s learning. But how easy is it to improve this (at scale) in poor countries? In a new CGD working paper we evaluate the impact of a large-scale school leader training programme impleme...
Blog Post
April 10, 2024
The UK Government has committed to producing a strategy on how the UK will support local leadership on development, climate, nature and humanitarian action; and its Foreign, Commonwealth and Development Office has endorsed a Donor Statement on Supporting Locally Led Development. Lisa Nandy, Labour’s...