Subscribe
Subscribe today to receive CGD’s latest newsletters and topic updates.
All Commentary
Filters:
Topics
Facet Toggle
Content Type
Facet Toggle
Blog Type
Facet Toggle
Time Frame
Facet Toggle
Blog Post
November 15, 2023
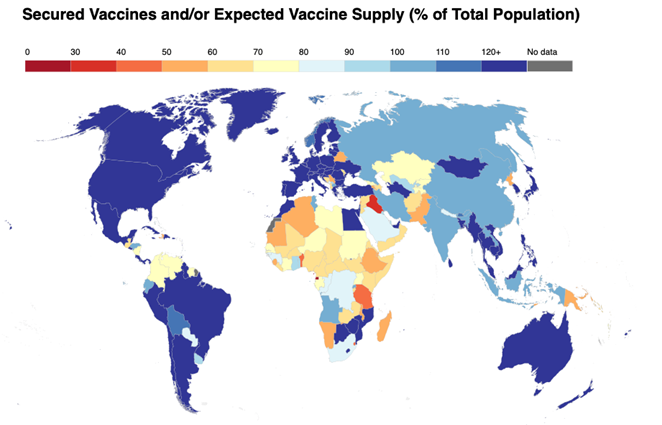
One of the most salient lessons from the pandemic is the need to diversify manufacturing capabilities and bolster supply resilience to avoid a repeat of the slow and inequitable vaccine roll out in African countries. To turn this lesson into action, the Gavi Secretariat is rapidly advancing plans fo...
Blog Post
February 15, 2022
Last week, the European Union and its member states placed a “European global health strategy” at the top of their shared agenda. And as part of the EU-Africa summit this week in Brussels, the EU is highlighting its support for regional manufacturing of COVID-19 vaccine via the African Union’s Partn...
Blog Post
August 12, 2021
This first episode of the new Africa-centered podcast series Lagos to Mombasa examines how African governments can shape their responses to the pandemic and prepare for the future. Patrick Tippoo of Biovac and the African Vaccine Manufacturing Initiative and Prashant Yadav from CGD join Gyude M...
Blog Post
February 10, 2021
In October 2020, the Center for Global Development—in partnership with African Union Development Agency-NEPAD (AUDA-NEPAD), UN Economic Commission for Africa (UNECA), and the African Leaders Malaria Alliance—hosted a high-level roundtable on regional health product manufacturing in Africa. This blog...
Blog Post
October 14, 2020
To maximise the impact of this badly needed investment to combat COVID and, most importantly, to avoid any perverse and potentially catastrophic implications of World Bank financing undermining current global efforts led by Gavi and CEPI, we propose that the World Bank commit to the four principles ...
Blog Post
January 15, 2020
Poor quality medicines pose significant risks to global health. This blog responds to a recent report from the National Academies that out clear recommendations to global, national and agency level stakeholders which can help strengthen food and medical products regulatory systems in low- and middle...