by
September 17, 2010
In a recent blog, I bemoaned the fact that donors were unable to secure the estimated $100 million needed to test and confirm the HIV prevention success of the CAPRISA microbicide. The comments on that blog by scientific writers Jon Cohen and Roger Tatoud point out that other trials of microbicide gels are in the works and that one in particular, the so-called VOICE trial, is both close to completion and particularly relevant.
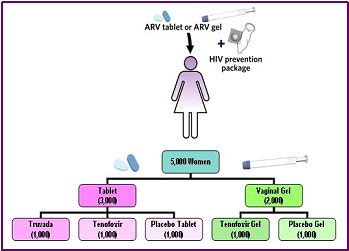
Because the VOICE trial is designed differently from the CAPRISA trial, it can only partly confirm CAPRISA. The VOICE trial is comparing the rate of HIV acquisition in approximately 5,000 HIV-negative African women who are randomized into one of five groups. In contrast to the women in the CAPRISA study, who were asked to use apply the microbicide gel only before and after sex, each group of women in the VOICE trial is asked to take an action every day for two years. The actions requested of the five groups are:
 Source: http://www.mtnstopshiv.org/news/studies/mtn003/qa
Success of either the oral or the vaginal method in the VOICE trial will depend on three unknowns: (1) the behavioral willingness of African women to conscientiously comply with the study directives every day for two years, (2) the biological efficacy of the product in relation to their frequency of use and (3) the degree to which daily exposure to these antiretroviral drugs can engender drug-resistant strains of HIV that can spread to others.
Other trials are in the works. For example, some researchers are hopeful that a silicon ring impregnated with the non nucleoside reverse transcriptase inhibitor dapivirine will be an even better option for women, one that they will only have to think about once a month when they replace the ring.
So, the question arises: Should donors continue to wait and see, hoping that studies of other ways of using microbicide gels will work much better than the CAPRISA approach? Are they right to wait for Godot – because technical progress is after all more certain than Godot’s arrival. Or should they scrape together the resources now to fund the studies to convince regulators to approve and policymakers to adopt the CAPRISA product?
Since the outcomes of future trials are unknown, there is no clear answer to this question. Any choice the donor makes will be a gamble. However, I still lean toward funding the confirmatory research and the development of the coitally-dependent approach that CAPRISA pioneered.
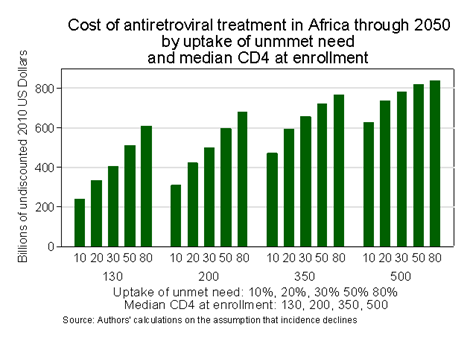
First, as I illustrate in the figure below, the amounts of money required to sustain and expand antiretroviral treatment entitlements are truly phenomenal. $200 billion between now and 2050 only pays for continuation of the current parsimonious trend of enrolling only 10 % of unmet need for treatment each year, where need is defined as it has been and the median CD4 count at recruitment is 130 cells per microliter. Offering Africans the kind of access to antiretroviral treatment available in the U.S. is likely to cost up to $800 billion through 2050. (Note from the graph that the cost of moving from 10 % to 80 % uptake is about half as great if enrollment is set to the high CD4 count of 500 than at the current median CD4 count of 130. This is because the newest version of my AIDSCost model incorporates the reduction in infectivity of those on treatment that has been observed in clinical trials.)
Source: http://www.mtnstopshiv.org/news/studies/mtn003/qa
Success of either the oral or the vaginal method in the VOICE trial will depend on three unknowns: (1) the behavioral willingness of African women to conscientiously comply with the study directives every day for two years, (2) the biological efficacy of the product in relation to their frequency of use and (3) the degree to which daily exposure to these antiretroviral drugs can engender drug-resistant strains of HIV that can spread to others.
Other trials are in the works. For example, some researchers are hopeful that a silicon ring impregnated with the non nucleoside reverse transcriptase inhibitor dapivirine will be an even better option for women, one that they will only have to think about once a month when they replace the ring.
So, the question arises: Should donors continue to wait and see, hoping that studies of other ways of using microbicide gels will work much better than the CAPRISA approach? Are they right to wait for Godot – because technical progress is after all more certain than Godot’s arrival. Or should they scrape together the resources now to fund the studies to convince regulators to approve and policymakers to adopt the CAPRISA product?
Since the outcomes of future trials are unknown, there is no clear answer to this question. Any choice the donor makes will be a gamble. However, I still lean toward funding the confirmatory research and the development of the coitally-dependent approach that CAPRISA pioneered.
First, as I illustrate in the figure below, the amounts of money required to sustain and expand antiretroviral treatment entitlements are truly phenomenal. $200 billion between now and 2050 only pays for continuation of the current parsimonious trend of enrolling only 10 % of unmet need for treatment each year, where need is defined as it has been and the median CD4 count at recruitment is 130 cells per microliter. Offering Africans the kind of access to antiretroviral treatment available in the U.S. is likely to cost up to $800 billion through 2050. (Note from the graph that the cost of moving from 10 % to 80 % uptake is about half as great if enrollment is set to the high CD4 count of 500 than at the current median CD4 count of 130. This is because the newest version of my AIDSCost model incorporates the reduction in infectivity of those on treatment that has been observed in clinical trials.)
 But a microbicide that is only 30% to 50% effective, in synergy with scaled up male circumcision and other prevention interventions, can save a big chunk of this money. Or looked at another way, the microbicide will reduce the cost of any given level of future treatment access. Greg and David, don’t you agree that improved prevention can help to make treatment affordable? That’s synergy between treatment and prevention, not opposition between them.
Second, African women will need to have several options. Just as in the case of birth control, where some women prefer the pill, some an injection, some condoms and some Plan B, women will not all choose the same method of HIV prevention. To me it seems likely that a coitally-dependent option will continue to find users in Africa, users who could not bring themselves to comply with a daily application or capsule and who are uncomfortable with the impregnated ring.
For these reasons, I think that the donors who have granted the entitlements are the ones who should step up to the plate here. Provided that those who propose spending $100 million can persuade others that this research is on the critical path to getting a coitally dependent microbicide gel licensed and into distribution in Africa within a few years, donors and advocates whose objective is expanded access to treatment should be the first to support this process. In the long run, only better HIV prevention will make expanded treatment affordable for all who need it.
Voltaire first pointed out that sometimes in human affairs [l]e mieux est l'ennemi du bien” (i.e. “the best is the enemy of the good”). Three centuries later, in Samuel Beckett’s play “Waiting for Godot,” two men are mired in indecision by the prospect that their savior might soon arrive – though he never does. I hope that donors currently hesitating to fund the CAPRISA replication studies will find a way to move a microbicide quickly toward regulatory approval – rather than waiting interminably for Godot.
But a microbicide that is only 30% to 50% effective, in synergy with scaled up male circumcision and other prevention interventions, can save a big chunk of this money. Or looked at another way, the microbicide will reduce the cost of any given level of future treatment access. Greg and David, don’t you agree that improved prevention can help to make treatment affordable? That’s synergy between treatment and prevention, not opposition between them.
Second, African women will need to have several options. Just as in the case of birth control, where some women prefer the pill, some an injection, some condoms and some Plan B, women will not all choose the same method of HIV prevention. To me it seems likely that a coitally-dependent option will continue to find users in Africa, users who could not bring themselves to comply with a daily application or capsule and who are uncomfortable with the impregnated ring.
For these reasons, I think that the donors who have granted the entitlements are the ones who should step up to the plate here. Provided that those who propose spending $100 million can persuade others that this research is on the critical path to getting a coitally dependent microbicide gel licensed and into distribution in Africa within a few years, donors and advocates whose objective is expanded access to treatment should be the first to support this process. In the long run, only better HIV prevention will make expanded treatment affordable for all who need it.
Voltaire first pointed out that sometimes in human affairs [l]e mieux est l'ennemi du bien” (i.e. “the best is the enemy of the good”). Three centuries later, in Samuel Beckett’s play “Waiting for Godot,” two men are mired in indecision by the prospect that their savior might soon arrive – though he never does. I hope that donors currently hesitating to fund the CAPRISA replication studies will find a way to move a microbicide quickly toward regulatory approval – rather than waiting interminably for Godot.
- Take a daily pill containing the antiretroviral drug tenofovir
- Take a daily pill containing both the antiretroviral drug tenofovir and the drug emtricitabine
- Take a daily pill containing only inactive ingredients like sugar (a placebo)
- Apply to the vagina a microbicide gel containing tenofovir
- Apply to the vagina a microbicide gel containing only inactive ingredients (a placebo)
 Source: http://www.mtnstopshiv.org/news/studies/mtn003/qa
Success of either the oral or the vaginal method in the VOICE trial will depend on three unknowns: (1) the behavioral willingness of African women to conscientiously comply with the study directives every day for two years, (2) the biological efficacy of the product in relation to their frequency of use and (3) the degree to which daily exposure to these antiretroviral drugs can engender drug-resistant strains of HIV that can spread to others.
Other trials are in the works. For example, some researchers are hopeful that a silicon ring impregnated with the non nucleoside reverse transcriptase inhibitor dapivirine will be an even better option for women, one that they will only have to think about once a month when they replace the ring.
So, the question arises: Should donors continue to wait and see, hoping that studies of other ways of using microbicide gels will work much better than the CAPRISA approach? Are they right to wait for Godot – because technical progress is after all more certain than Godot’s arrival. Or should they scrape together the resources now to fund the studies to convince regulators to approve and policymakers to adopt the CAPRISA product?
Since the outcomes of future trials are unknown, there is no clear answer to this question. Any choice the donor makes will be a gamble. However, I still lean toward funding the confirmatory research and the development of the coitally-dependent approach that CAPRISA pioneered.
First, as I illustrate in the figure below, the amounts of money required to sustain and expand antiretroviral treatment entitlements are truly phenomenal. $200 billion between now and 2050 only pays for continuation of the current parsimonious trend of enrolling only 10 % of unmet need for treatment each year, where need is defined as it has been and the median CD4 count at recruitment is 130 cells per microliter. Offering Africans the kind of access to antiretroviral treatment available in the U.S. is likely to cost up to $800 billion through 2050. (Note from the graph that the cost of moving from 10 % to 80 % uptake is about half as great if enrollment is set to the high CD4 count of 500 than at the current median CD4 count of 130. This is because the newest version of my AIDSCost model incorporates the reduction in infectivity of those on treatment that has been observed in clinical trials.)
Source: http://www.mtnstopshiv.org/news/studies/mtn003/qa
Success of either the oral or the vaginal method in the VOICE trial will depend on three unknowns: (1) the behavioral willingness of African women to conscientiously comply with the study directives every day for two years, (2) the biological efficacy of the product in relation to their frequency of use and (3) the degree to which daily exposure to these antiretroviral drugs can engender drug-resistant strains of HIV that can spread to others.
Other trials are in the works. For example, some researchers are hopeful that a silicon ring impregnated with the non nucleoside reverse transcriptase inhibitor dapivirine will be an even better option for women, one that they will only have to think about once a month when they replace the ring.
So, the question arises: Should donors continue to wait and see, hoping that studies of other ways of using microbicide gels will work much better than the CAPRISA approach? Are they right to wait for Godot – because technical progress is after all more certain than Godot’s arrival. Or should they scrape together the resources now to fund the studies to convince regulators to approve and policymakers to adopt the CAPRISA product?
Since the outcomes of future trials are unknown, there is no clear answer to this question. Any choice the donor makes will be a gamble. However, I still lean toward funding the confirmatory research and the development of the coitally-dependent approach that CAPRISA pioneered.
First, as I illustrate in the figure below, the amounts of money required to sustain and expand antiretroviral treatment entitlements are truly phenomenal. $200 billion between now and 2050 only pays for continuation of the current parsimonious trend of enrolling only 10 % of unmet need for treatment each year, where need is defined as it has been and the median CD4 count at recruitment is 130 cells per microliter. Offering Africans the kind of access to antiretroviral treatment available in the U.S. is likely to cost up to $800 billion through 2050. (Note from the graph that the cost of moving from 10 % to 80 % uptake is about half as great if enrollment is set to the high CD4 count of 500 than at the current median CD4 count of 130. This is because the newest version of my AIDSCost model incorporates the reduction in infectivity of those on treatment that has been observed in clinical trials.)
 But a microbicide that is only 30% to 50% effective, in synergy with scaled up male circumcision and other prevention interventions, can save a big chunk of this money. Or looked at another way, the microbicide will reduce the cost of any given level of future treatment access. Greg and David, don’t you agree that improved prevention can help to make treatment affordable? That’s synergy between treatment and prevention, not opposition between them.
Second, African women will need to have several options. Just as in the case of birth control, where some women prefer the pill, some an injection, some condoms and some Plan B, women will not all choose the same method of HIV prevention. To me it seems likely that a coitally-dependent option will continue to find users in Africa, users who could not bring themselves to comply with a daily application or capsule and who are uncomfortable with the impregnated ring.
For these reasons, I think that the donors who have granted the entitlements are the ones who should step up to the plate here. Provided that those who propose spending $100 million can persuade others that this research is on the critical path to getting a coitally dependent microbicide gel licensed and into distribution in Africa within a few years, donors and advocates whose objective is expanded access to treatment should be the first to support this process. In the long run, only better HIV prevention will make expanded treatment affordable for all who need it.
Voltaire first pointed out that sometimes in human affairs [l]e mieux est l'ennemi du bien” (i.e. “the best is the enemy of the good”). Three centuries later, in Samuel Beckett’s play “Waiting for Godot,” two men are mired in indecision by the prospect that their savior might soon arrive – though he never does. I hope that donors currently hesitating to fund the CAPRISA replication studies will find a way to move a microbicide quickly toward regulatory approval – rather than waiting interminably for Godot.
But a microbicide that is only 30% to 50% effective, in synergy with scaled up male circumcision and other prevention interventions, can save a big chunk of this money. Or looked at another way, the microbicide will reduce the cost of any given level of future treatment access. Greg and David, don’t you agree that improved prevention can help to make treatment affordable? That’s synergy between treatment and prevention, not opposition between them.
Second, African women will need to have several options. Just as in the case of birth control, where some women prefer the pill, some an injection, some condoms and some Plan B, women will not all choose the same method of HIV prevention. To me it seems likely that a coitally-dependent option will continue to find users in Africa, users who could not bring themselves to comply with a daily application or capsule and who are uncomfortable with the impregnated ring.
For these reasons, I think that the donors who have granted the entitlements are the ones who should step up to the plate here. Provided that those who propose spending $100 million can persuade others that this research is on the critical path to getting a coitally dependent microbicide gel licensed and into distribution in Africa within a few years, donors and advocates whose objective is expanded access to treatment should be the first to support this process. In the long run, only better HIV prevention will make expanded treatment affordable for all who need it.
Voltaire first pointed out that sometimes in human affairs [l]e mieux est l'ennemi du bien” (i.e. “the best is the enemy of the good”). Three centuries later, in Samuel Beckett’s play “Waiting for Godot,” two men are mired in indecision by the prospect that their savior might soon arrive – though he never does. I hope that donors currently hesitating to fund the CAPRISA replication studies will find a way to move a microbicide quickly toward regulatory approval – rather than waiting interminably for Godot.DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.





