Subscribe
Subscribe today to receive CGD’s latest newsletters and topic updates.
All Commentary
Filters:
Topics
Facet Toggle
Content Type
Facet Toggle
Blog Type
Facet Toggle
Time Frame
Facet Toggle
Blog Post
April 18, 2024
Achieving the UN Sustainable Development Goals (SDGs) and addressing global challenges will require a step change in private investment in emerging markets and developing economies (EMDEs). Only a small fraction of the trillions in private assets under management are currently directed to EMDEs.
Blog Post
April 17, 2024
In the wake of COVID and amidst global crises, care has increasingly become recognized as a global issue critical for sustainable development and gender equality. Yet, there are still massive gaps in care policies, services, and financing, and there is much more work that needs to happen to ensure u...
Blog Post
April 16, 2024
Last month The Gambia’s National Assembly advanced a bill that, if ratified, would make it the first country to overturn a ban on female genital mutilation. These moves—supported by the predominantly male legislature—reflect the precarious nature of gains made in gender equality and have implication...
Blog Post
April 15, 2024
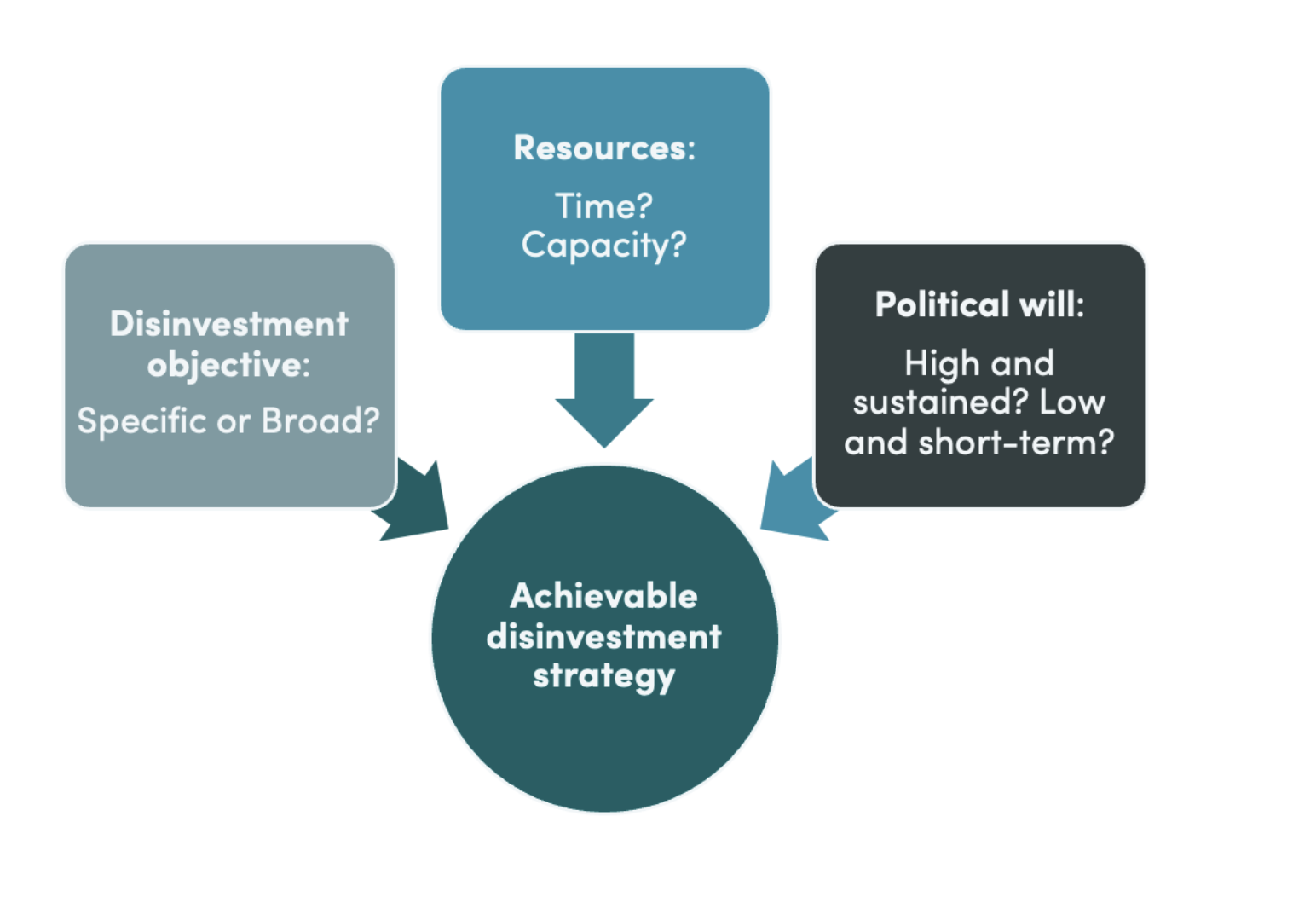
In theory, disinvestment offers the potential to release resources for achieving UHC during challenging fiscal times. However, in practice, it is time- and resource-consuming and results in less benefit than expected at the outset. The opportunity cost of launching an initiative needs to be weighed ...
Blog Post
April 11, 2024
There are currently two prevailing methodologies for calculating PCM—the MDB approach and the OECD approach. Both approaches have developed over time and contributed significantly to better understanding of the ways in which private capital is mobilised for development. However, they have key defici...