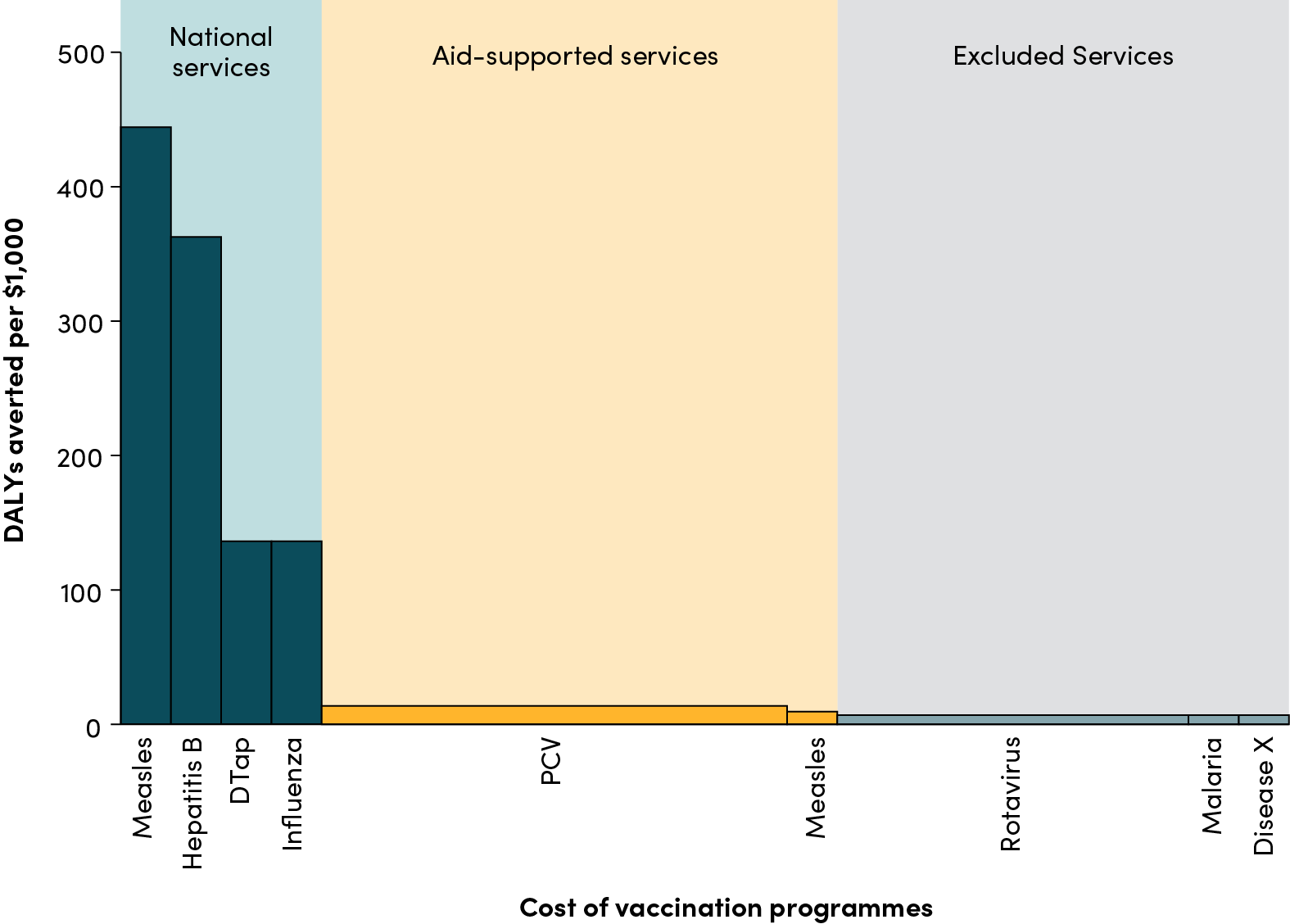
Victoria Hale, head of OneWorld Health, an innovative non-profit pharmaceutical firm, reckons that compulsory licensing could prove "the last blow" that pushes the drug industry away from looking for cures for diseases of the poor world, which are already woefully neglected...Bruce Lehman, a lawyer who worked on the TRIPS [sic] accord in the Clinton administration, thinks it is cynical for middle-income countries "to avoid paying their fair share of drug-discovery costs."
The Economist, June 7, 2007The recent decisions by Thailand and Brazil to issue compulsory licenses for AIDS drugs, and in Thailand's case a heart-disease drug, triggered strong responses from around the world - anger from drug companies but support from many development NGOs. Often, however, one finds more heat than light reflected in this debate over intellectual property rights and access to drugs in developing countries and it is useful to keep in mind both what we know and what we don't.What we know:First, socially optimal levels of protection for intellectual property rights (IPRs) always represent a balance between the need to provide incentives for innovation and the social benefits of disseminating the fruits of innovative activity, for example new and better drugs, as widely as possible. Poor countries with little or no innovative activity gain little and can even lose from enforcing strong IPRs. As reflected in the article in a quote from an official of Ranbaxy, an Indian drug company, as innovative activity increases, developing countries do gain from strong IP protection and can be expected to enforce those laws more effectively.Second, developing countries are fully within their rights under the World Trade Organization agreement on IPRs when issuing compulsory licenses in the case of national health emergencies and AIDS has been recognized as such an emergency. Third, very little R&D spending is devoted to tropical and other diseases that primarily affect developing countries. Although dated, a World Health Organization study estimated that only 4 percent of global public and private R&D spending "was related to health problems of low- and middle-income countries" in 1992 (cited in a
CGD Working Paper by Jenny Lanjouw). A more recent analysis by the
Global Forum for Health Research estimates that 90 percent of research funds are spent studying diseases that affect just 10 percent of the world's population. Stronger IPRs will not change this situation because these countries are simply too small and too poor to generate the returns required to cover the R&D costs. Still, there is much that we do not know. How important are the middle-income countries as a source of profits for multinational drug companies? What is a "fair share" for them to pay and for which drugs? In addition to
Advance Market Commitments, are there other mechanisms that could be developed to stimulate research into diseases that affect primarily developing countries? Chronic and noncommunicable "rich-world" diseases, such as diabetes and heart disease, are growing problems in developing countries but these are not generally regarded as constituting "emergencies" or as posing global health threats (like avian flu). For many common diseases that have drug therapies, are patents the major obstacles to access? One
analysis (.pdf) of WHO's Model List of Essential Medicines finds that only 17 of 319 items are patentable and that many of those are not under patent.There are no easy answers to these questions. But the aim of the Center is to continue to analyze them in a rigorous way in hopes of shifting the balance in the debate from heat to light.
CGD blog posts reflect the views of the authors, drawing on prior research and experience in their areas of expertise.
CGD is a nonpartisan, independent organization and does not take institutional positions.