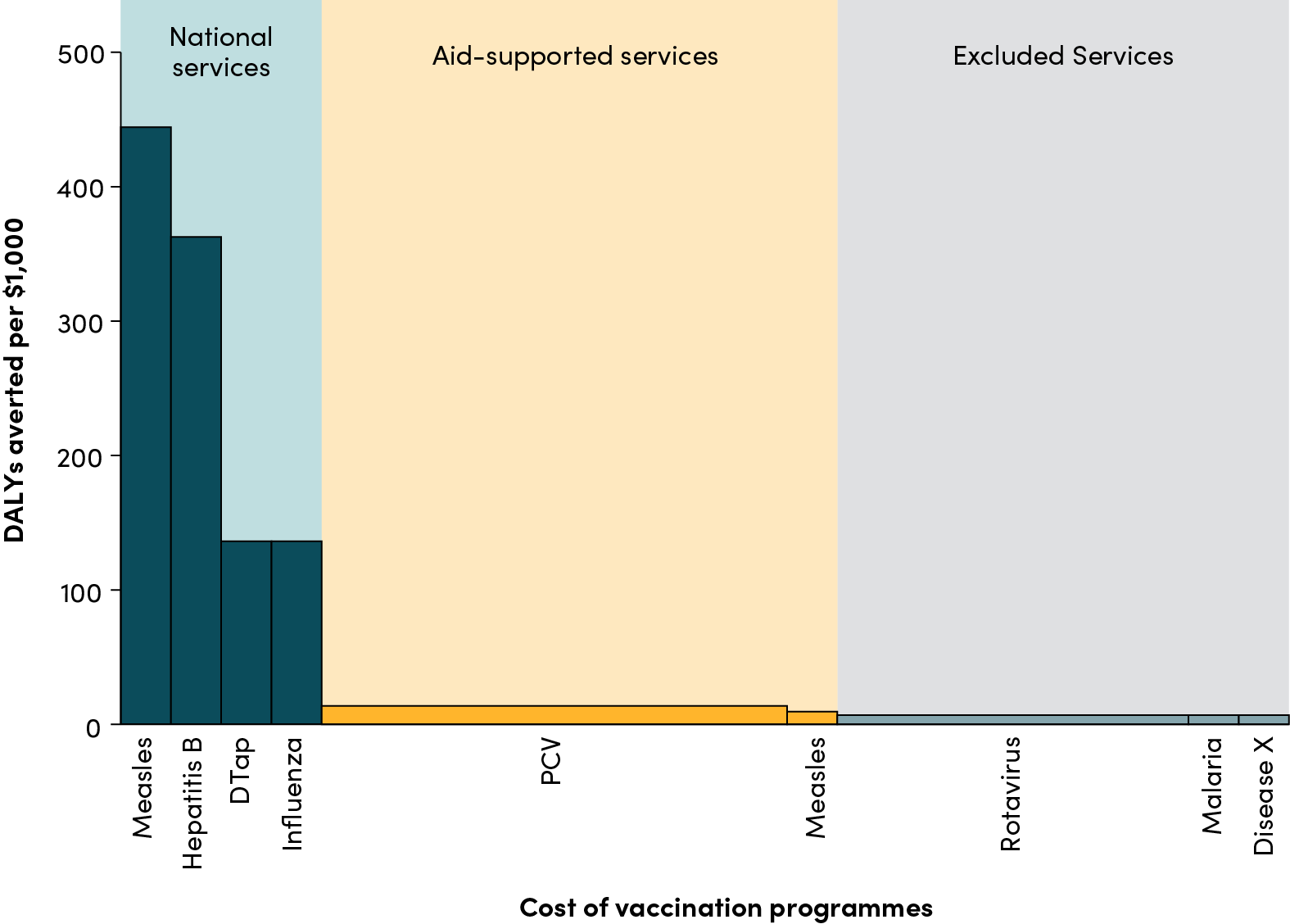
Even though the TRIPS agreement and the subsequent Doha declaration contain public health safeguards that allow developing countries to produce or import generic medicines, the reality is that few countries have had the political will and technical support to use those safeguards. That's why it's especially encouraging that earlier this week, Thailand's government announced it would issue a compulsory license for the antiretroviral drug Efavirenz, currently patented there by Merck. The drug will initially be imported from India, but local production by the Government Pharmaceutical Organization would begin next year. If issued, the compulsory license would have the effect of halving the price of the drug, from roughly $40/month to $20 - a significant development in a country with nearly 600,000 people living with HIV. As a recent World Bank report has shown, the sustainability of the Thai government’s successful AIDS program depends crucially on the availability of cheaper first-line and second-line ARVs.
The much broader significance of the Thai government"s decision, however, is that it could set an important precedent whereby other developing countries facing public health crises would be emboldened to take advantage of TRIPS safeguards such as compulsory licensing. These actions could potentially expand the market for many important pharmaceuticals - not only ARVs - and increase competition among producers. A number of second-line ARVs are currently not available generically in developing countries; compulsory licensing would be a solution in some cases.
In the past, patent-holding firms that face a compulsory licensing threat have often responded by immediately reducing the price of the drug at stake or issuing a voluntary license for a single generic producer from the patent-holder (South Africa and Brazil, for example, have experience here). The same thing seems to be happening in Thailand now, with the Financial Times reporting that Merck has offered to slash its price for efavirenz. It would be unfortunate if this keeps the Thai government from proceeding with the compulsory license - competition among producers in developing countries with manufacturing capacity is critical if we want prices there to be as low as possible.
CGD blog posts reflect the views of the authors, drawing on prior research and experience in their areas of expertise.
CGD is a nonpartisan, independent organization and does not take institutional positions.